What a New Study Gets Wrong About Fish Oil
And why association is not causation
Today is Mother's Day — and for many of us, the fear of watching someone we love lose their memory is very real. Wishing all mothers a beautiful day.
A new study published in the Journal of Prevention of Alzheimer’s Disease made headlines with a finding that seems to defy common sense: people who took omega-3 supplements in the Alzheimer’s Disease Neuroimaging Initiative (ADNI) cohort showed faster cognitive decline than those who didn’t. If true, should millions of people stop taking their fish oil capsules?
Not so fast. While the findings deserve attention, understanding what this study can and cannot tell us is essential before drawing any conclusions.
What the Study Found
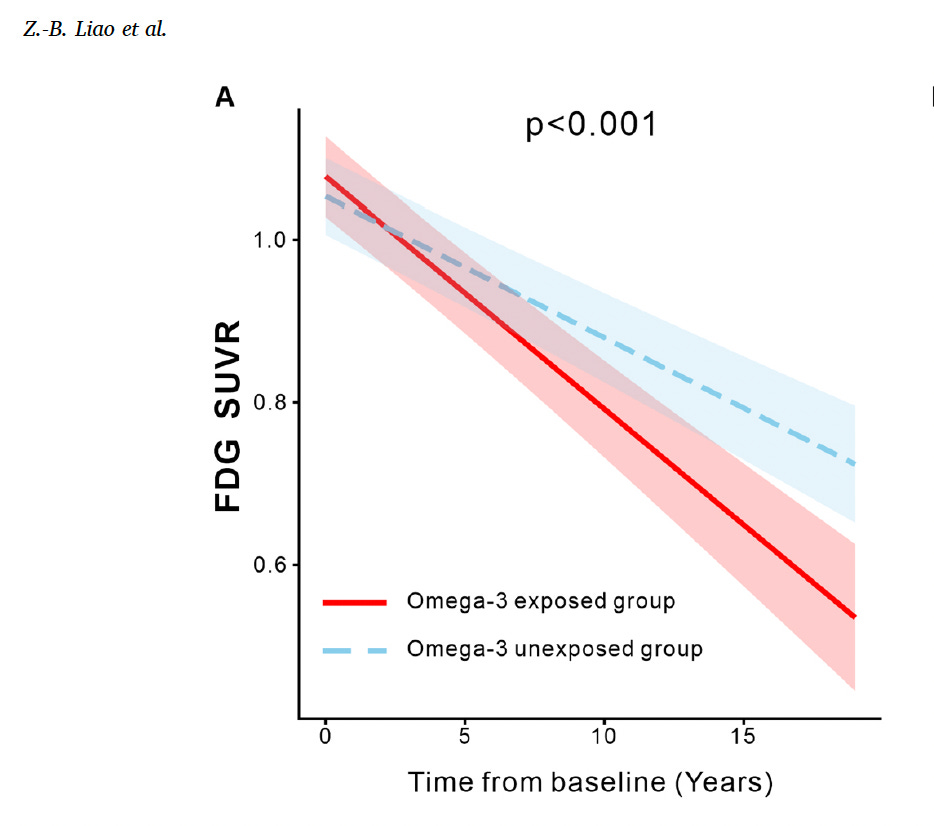
The researchers analyzed data from 819 older adults in the ADNI database, comparing 273 omega-3 users — primarily fish oil — to 546 matched non-users over a median of five years. Using statistical techniques designed to create comparable groups (propensity score matching), they found that omega-3 users showed faster declines on three standard cognitive tests: the MMSE, ADAS-Cog13, and CDR-SB.
Importantly, this accelerated decline appeared linked not to the classic hallmarks of Alzheimer’s disease — amyloid plaques or tau tangles — but to reduced glucose metabolism in key brain regions, a marker of synaptic dysfunction or damaged mitochondria where omega-3s may get oxidized. The authors proposed that commercially available fish oil, which is particularly vulnerable to oxidation, might be generating harmful byproducts that damage synaptic function. It’s a provocative hypothesis. But the study design makes it very difficult to draw that conclusion.
This figure is striking visually, but it illustrates the core limitation — an observational design cannot tell us whether omega-3s caused the faster metabolic decline, or whether people already heading toward faster decline were the ones who reached for supplements.
The Problem with Observational Studies
The ADNI analysis is an observational study — it watches what people do in real life and looks for patterns. This is a valuable starting point, but it comes with a fundamental limitation: people who take supplements are systematically different from those who don’t, and no amount of statistical adjustment can perfectly account for all those differences.
The authors used propensity score matching, a technique that pairs omega-3 users with non-users who look similar on measured characteristics like age, sex, APOE genetic status, and diagnosis. But “similar on paper” is not the same as “truly comparable.” People who reach for a fish oil bottle may do so because a physician flagged concerns about their cognition, because they are managing cardiovascular or inflammatory conditions, or because they’ve noticed early memory changes themselves. These hidden motivations — what researchers call unmeasured confounders — can create a spurious association between a supplement and a bad outcome, even when the supplement itself is entirely innocent.
There is also a well-known phenomenon in observational research called “healthy user bias,” where supplement takers tend to be healthier overall than non-takers. In this case, however, the study found the opposite — omega-3 users appeared to do worse. The authors acknowledge this paradox themselves, noting that it “presents a puzzle” since healthy user bias would typically favor detecting a protective effect. This unusual direction of effect makes unmeasured confounding an especially important concern.
Propensity Scoring Does Not Eliminate Reverse Causation
This brings us to one of the most critical points in interpreting this kind of research: propensity score matching reduces baseline imbalances on measured variables, but it cannot solve the problem of reverse causation. That is, people may begin taking omega-3 supplements because they are already experiencing subtle cognitive concerns — concerns that haven’t yet shown up on formal tests but are real enough to prompt self-medication.
The authors tried to address this by comparing cognitive trajectories during the pre-supplementation period, finding no significant differences. That’s a thoughtful step. But it is an incomplete test. Pre-clinical Alzheimer’s pathology — the kind that silently accumulates for years before symptoms emerge — can generate subtle functional concerns that motivate lifestyle changes long before they register on cognitive scales. No retrospective analysis of pre-treatment trajectories can fully rule out this possibility. The authors themselves noted that their analysis “may miss subtle trajectories that prompt individuals to start supplements.” That is not a minor caveat — it is the central interpretive challenge.
Putting It in Context: What the Rest of the Evidence Says
Perhaps the most important corrective to this study is the broader evidence base. In the UK Biobank — one of the largest prospective cohorts ever assembled, following hundreds of thousands of participants — omega-3 supplement use was associated with lower rates of dementia, not higher. If omega-3 supplements were genuinely accelerating neurodegeneration, we would expect to see that signal consistently across different populations and methodologies. We don’t.
Large randomized controlled trials, which are far less vulnerable to confounding, have consistently found something different and more mundane: omega-3 supplements produce no significant benefit in people with established Alzheimer’s disease or mild cognitive impairment, but they also cause no meaningful harm. Multiple meta-analyses have reached similar conclusions. “Ineffective” and “harmful” are very different verdicts, and the RCT evidence supports the former, not the latter.
The ADNI findings may also reflect a quirk of the supplement itself. Studies have found that a large proportion of commercially available fish oil products are already oxidized before they are consumed — meaning the capsule delivers not clean DHA and EPA, but a mixture that includes pro-inflammatory breakdown products. If the ADNI signal reflects anything real, it may tell us more about supplement quality than about omega-3 biology.
Why Omega-3 Supplements May Have Only a Limited Role
Even setting aside these methodological concerns, there are strong reasons to think that omega-3 supplements taken in isolation may simply not be potent enough to meaningfully alter Alzheimer’s trajectories in either direction. The biology is more complicated than the supplement industry suggests.
DHA — the primary omega-3 fatty acid in the brain — has genuine neuroprotective functions: maintaining membrane fluidity, supporting synaptic signaling, resolving inflammation, and facilitating amyloid clearance. But how effectively a supplement translates into actual brain DHA levels depends heavily on individual factors: APOE genetic status, age, existing brain pathology, baseline omega-3 levels, and the health of the gut microbiome. In people with already-established Alzheimer’s pathology — where glucose metabolism is disrupted, oxidative stress is high, and the blood-brain barrier is compromised — the situation becomes even more complex. Under these conditions, additional DHA may face a hostile metabolic environment, and any benefit is easily overwhelmed.
This is not a reason to conclude that omega-3s are harmful. It is a reason to conclude that supplements, delivered as isolated capsules outside the context of overall dietary patterns, are likely to have a modest — and possibly negligible — effect on cognitive aging.
There is also a little-known biological wrinkle: high-dose DHA supplementation can paradoxically suppress the body’s own DHA production. Research published by the Bazinet group demonstrates that dietary DHA inhibits a liver enzyme called ELOVL2, which elongates EPA along the DHA biosynthesis pathway. In plain terms, when you take a DHA supplement, you send a signal to your liver to stop making its own DHA from plant-based precursors found in foods like flaxseed and walnuts. The body is tuned for a steady dietary supply — not a daily bolus from a capsule — and flooding it with preformed DHA suppresses the more nuanced, on-demand synthesis the liver would otherwise perform. Long-term supplementation may therefore not simply add DHA on top of what the body makes; it may partly replace endogenous production with an external dependency.
Compounding this is the problem of oxidation. DHA and EPA are polyunsaturated fats — chemically fragile and prone to oxidative damage. When the metabolic environment is already compromised, as in an aging brain facing elevated oxidative stress, disrupted glucose metabolism, and mitochondrial dysfunction, supplemental omega-3s may not arrive at their destination intact. Rather than being incorporated into neuronal membranes to support synaptic function, they can be oxidized into pro-inflammatory byproducts. This is why the context in which omega-3s are consumed may matter as much as the omega-3s themselves. A healthy dietary and lifestyle pattern — regular physical activity, a diet rich in fiber and polyphenols, restorative sleep, metabolic health — creates the cellular conditions under which DHA can actually be used well. The same fatty acids delivered into a dysfunctional metabolic environment may have a very different fate.
Uncertainty and Future Directions
There is much we genuinely do not know. Whether omega-3 supplementation could prevent Alzheimer’s if started early enough — potentially decades before symptoms — in people who are genetically vulnerable and nutritionally deficient remains an open question. The PreventE4 trial — testing high-dose DHA in cognitively normal APOE4 carriers well before the onset of cognitive decline — is expected to publish its results within the next two months. If high-dose DHA shows meaningful benefit in this genetically at-risk, presymptomatic population, it would reframe the entire debate: not whether omega-3s work, but when and for whom the intervention needs to start. We also don’t understand well how the gut microbiome shapes the response to omega-3 supplementation, or whether certain genetic profiles (such as APOE4 carriers with low baseline omega-3 levels) might benefit more than others.
Supplement quality is also an underappreciated problem. If fish oil products are frequently oxidized by the time of consumption, then what we are testing in many observational studies is not omega-3s per se, but a degraded product. Rigorous studies with verified, high-quality formulations are needed.
Finally, the broader question remains unresolved: do omega-3s work better as part of a complex dietary pattern than as a standalone supplement? Emerging evidence consistently suggests yes. Whole omega-3 dietary patterns — both of which feature regular fatty fish consumption alongside fiber, polyphenols, and other anti-inflammatory nutrients — show more consistent associations with cognitive resilience than any single nutrient taken in isolation. The whole appears to be greater than the sum of its parts.
Take-Home Messages
The ADNI finding is thought-provoking but should not cause alarm. Observational studies cannot establish causation, and the pattern seen here — where omega-3 users appear sicker than non-users — is more consistent with reverse causation than with genuine neurotoxicity. The UK Biobank and randomized trial evidence do not support the conclusion that fish oil harms the brain.
Omega-3 supplements are unlikely to have a major effect in either direction for most people. The balance of evidence suggests that they don’t meaningfully slow Alzheimer’s disease in people who already have cognitive impairment or established pathology, but they also don’t accelerate it. The story is mostly one of modest-to-neutral impact.
A diet naturally rich in omega-3s is almost certainly more beneficial than any supplement. Eating fatty fish regularly, or omega-3s from plants, alongside a diet high in vegetables, fiber, and plant-based foods, delivers omega-3s in an unoxidized form, within a matrix of other nutrients, in a gut environment that supports beneficial metabolism. That package — not a capsule — is what the observational evidence consistently associates with better brain aging.
Don’t throw away your fish oil based on a single observational study. But do invest more in your overall diet. No supplement replaces the complexity of real food, and this study — whatever its ultimate interpretation — is a useful reminder of that.
References
Liao et al. “The association between omega-3 supplementation and cognitive decline in older adults.” Journal of Prevention of Alzheimer’s Disease, 2026.
Metherel AH et al. “Dietary docosahexaenoic acid (DHA) downregulates liver DHA synthesis by inhibiting eicosapentaenoic acid elongation.” Journal of Lipid Research, 65(6):100548, 2024.
Huang Y et al. "Associations of fish oil supplementation with incident dementia: Evidence from the UK Biobank cohort study." Frontiers in Neuroscience, 2022.
Kerman BE, Self WK, Yassine HN. “Can the gut microbiome inform the effects of omega-3 supplementation?” Current Opinion in Clinical Nutrition and Metabolic Care, 2023.
Ebright B, Duro MV, Chen J, Louie G, Yassine HN. “APOE4 effects on DHA brain metabolism across the Alzheimer’s disease spectrum.” Trends in Endocrinology & Metabolism, 2024.



I will dedicate a post on this in the next month or two, when we get PreventE4 trial out. It is coming soon!
Super helpful.