Red Meat, APOE4, and the Limits of Observational Nutrition Science
Observational studies can drive you nuts!
Last week, a science reporter reached out asking me to comment on a new paper in JAMA Network Open showing that APOE4 carriers may have a different relationship with meat consumption than the rest of the population. She wanted a quote. I owe her an apology.
The honest truth is that academic life had swallowed me whole at the time. Grant deadlines, manuscript revisions, back-to-back committee meetings, study section, a clinical schedule that doesn’t pause for anyone. I saw her message, thought I need to think carefully about this one. So belatedly, here is my take:
What the Study Did and Found
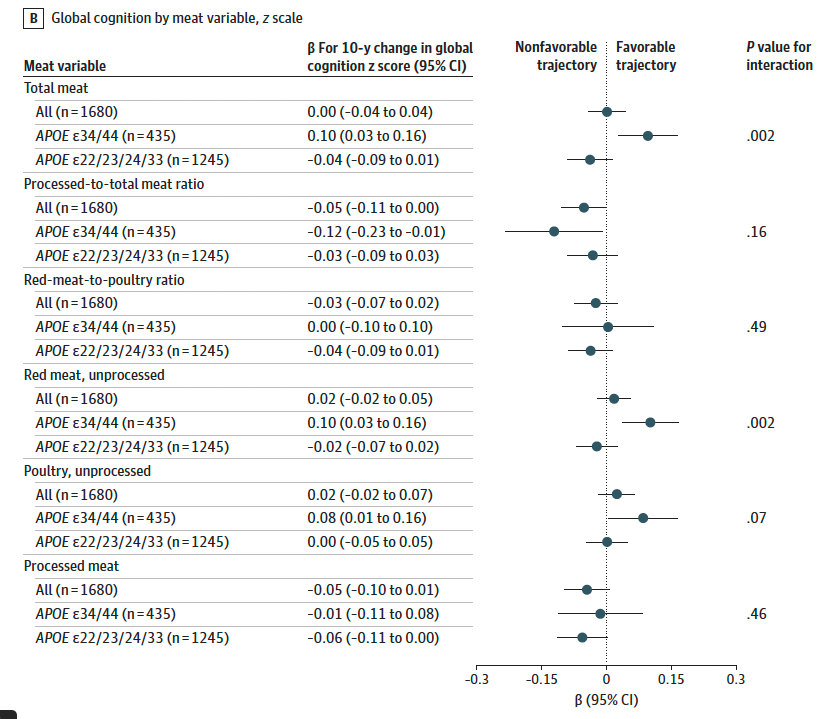
“Meat Consumption and Cognitive Health by APOE Genotype” by Norgren, Carballo-Casla, and colleagues at Karolinska Institutet is a prospective cohort study published March 19, 2026 in JAMA Network Open, drawing from the Swedish National Study on Aging and Care, Kungsholmen (SNAC-K). Of 5,111 randomly selected adults aged 60 and older, 2,157 met inclusion criteria — no dementia at baseline, with available diet, cognition, and APOE data. Mean age was 71.2 years; 62% were women; 569 participants (26.4%) carried APOE ε3/ε4 or ε4/ε4 genotypes (APOE34/44). Follow-up extended up to 15 years, during which 296 participants developed dementia.
Dietary exposure was assessed using a validated 98-item food frequency questionnaire at baseline and at 3- and 6-year follow-ups, with total meat consumption expressed as grams per total kilocalories. Secondary exposures included the ratio of processed to total meat and the log ratio of unprocessed red meat to poultry. Covariates were selected using causal inference principles and included age, sex, education, physical activity, smoking, alcohol, total energy intake, morbidity, baseline cognition, and — methodologically important — the Alternative Healthy Eating Index (AHEI) score excluding meat items, ensuring the association was not simply a marker of overall dietary quality.
Cognitive trajectories (composite z-score across episodic memory, semantic memory, verbal fluency, and perceptual speed over 15 years, available in 1,680 participants) were analyzed by linear regression with quintile-based exposure. Dementia incidence was analyzed using Fine and Gray subdistribution hazard ratios (sHRs), treating non-dementia death as a competing risk. To address reverse causation, the authors used a triangulation approach — combining longitudinal between-participant, within-participant fixed-effects, and cross-sectional baseline analyses — and confirmed that dietary changes did not differ between those who did and did not develop dementia.
Main findings: in APOE34/44 carriers, the top versus bottom quintile of meat intake was associated with better cognitive trajectories (β = 0.32; 95% CI, 0.07–0.56; P = .01), most pronounced for episodic memory (β = 0.52; 95% CI, 0.12–0.92). No association was found in non-carriers (β = −0.11; P = .20). The APOE interaction on cognitive trajectory: P = .004. For dementia, the sHR in APOE34/44 carriers was 0.45 (95% CI, 0.21–0.95; P = .04) versus 0.95 in non-carriers — but the APOE-by-meat interaction on dementia did not reach statistical significance (P = .10). Processed meat increased dementia risk regardless of genotype (sHR, 1.14; 95% CI, 1.01–1.29, Figure below). Post-hoc analyses suggested a consistent APOE interaction for all-cause mortality (HR, 0.85; P = .04) and greater vitamin B12 absorption from meat in APOE34/44 carriers.
Does P = .10 for the Dementia Interaction Undermine the Whole Paper?
This is the right question to ask, and it deserves a direct answer.
The P = .10 for the APOE-by-meat interaction applies specifically to the dementia diagnosis endpoint. With only 569 APOE34/44 participants and 296 total dementia cases across all genotypes, the study was underpowered to reliably detect a genotype interaction for incident dementia — this is a harder, rarer endpoint than a continuously measured cognitive score. The directionality of the dementia finding is fully consistent with the cognitive trajectory result; it simply does not clear the significance threshold for that specific secondary outcome.
The paper’s primary pre-specified outcome was cognitive trajectory, analyzed in 1,680 participants with 15 years of longitudinal data, where the interaction is P = .004 and survives multiple sensitivity analyses including participants with higher HbA1c, lower AHEI scores, and higher vascular risk scores. The triangulation approach yielded consistent results across three analytical methods. The dementia result is best understood as directionally supportive but statistically inconclusive — not as evidence against the cognitive finding.
That said, the P = .10 is a legitimate reason for interpretive caution. The paper supports the hypothesis that APOE genotype modifies the association between meat consumption and cognitive aging trajectories in a Swedish elderly cohort. That is what it shows — not that red meat prevents Alzheimer’s disease.
Why This Paper Is Still Worth Taking Seriously
Beyond the pre-specified hypothesis and triangulation design, several features add credibility. The AHEI adjustment (excluding meat) means the cognitive signal is not merely reflecting that high meat consumers also eat more vegetables and fish. The effect was robust among women, participants under 72, those with higher HbA1c, and those with higher cardiovascular risk scores — subgroups where confounding by general healthiness is less likely. Post-hoc findings for mortality and B12 absorption point in the same direction. And the distinction between processed and unprocessed meat — with processed meat conferring risk regardless of genotype — is internally coherent and consistent with the broader literature.
The limitations are real: self-reported diet, a single urban Swedish cohort with limited generalizability across ancestries, a modest APOE34/44 subgroup, and the inherent inability of observational data to establish causality despite the methodological care. The study is best received as a well-grounded hypothesis-generating contribution that makes a compelling case for APOE-stratified randomized trials.
The Biology: Why APOE4 Carriers May Respond Differently to Dietary Fat
The observational finding has a plausible biological basis, though the mechanism points somewhere more specific than “APOE4 carriers need more fat.”
In a review I wrote with Caleb “Tuck” Finch in Frontiers in Aging Neuroscience, we examined what apoE4’s cell biology implies for brain energy metabolism. APOE4 impairs the brain’s ability to use glucose efficiently — APOE4 astrocytes have roughly half the glucose uptake capacity of APOE3 astrocytes, and neuroimaging confirms regional brain glucose hypometabolism in APOE4 carriers detectable decades before symptoms. In response, the APOE4 brain shifts toward dietary fat as its preferred energy source. This may be one reason why older APOE4 carriers who lose significant weight show accelerated cognitive decline — adipose fat stores appear to serve as an important fuel reservoir for a brain that can no longer rely as heavily on glucose.
This energy-preference hypothesis has direct clinical support. Hanson and colleagues (2015) showed in a blinded feeding study that an acute high-fat meal improved cognitive performance and plasma Alzheimer’s disease biomarkers in APOE4 carriers with mild cognitive impairment — while non-carriers showed the opposite pattern. Fat, in a glucose-deprived APOE4 brain, appears to be a better fuel.
This provides a biologically coherent explanation for the SNAC-K finding: if the APOE4 brain runs better on fat, a dietary pattern that includes more fat — including from unprocessed meat — may support cognitive function in ways that do not apply to non-carriers.
The important qualification: the APOE4 brain’s preference for fat does not translate into a recommendation to eat more red meat or to adopt a ketogenic diet. The fat that matters most for the APOE4 brain is high-quality polyunsaturated fat, particularly the omega-3 fatty acid DHA. Using PET imaging, we showed that brain DHA uptake is approximately 20% greater in younger cognitively normal APOE4 carriers versus non-carriers — a signal of heightened biological demand. That demand is best met through fish and seafood. Red meat, particularly unprocessed, may contribute to overall fat and protein intake in ways that modestly support this, but it is not the primary source, and processed red meat — high in sodium, nitrates, and additives — increases dementia risk across all genotypes and should be minimized regardless of APOE status.
Dietary Patterns, Not Individual Foods
The most important and most frequently overlooked principle in nutrition and dementia research is that the relevant unit of analysis is a dietary pattern, not a single food. In our Lancet Healthy Longevity paper, we reviewed the full evidence base — observational studies, clinical trials, and mechanistic data — and found that no individual nutrient or food has demonstrated consistent cognitive benefit in adequately powered randomized trials when evaluated in isolation. The Mediterranean and MIND dietary patterns show the most consistent associations with cognitive health, not because of any single component but because of their structural coherence: high vegetables, whole grains, legumes, fish, olive oil, moderate unprocessed animal protein, and minimal processed food and added sugar. The SNAC-K finding that processed meat increases dementia risk regardless of genotype fits squarely within this framework. For APOE4 carriers, the most evidence-supported approach is a pattern that provides adequate omega-3 fatty acids, preserves insulin sensitivity, and minimizes ultra-processed foods — not one defined by a target intake of any single protein source.
There is, however, a complication in applying Mediterranean diet evidence to the SNAC-K cohort that is worth naming. Sweden is not a Mediterranean country, and long-term adherence to a Mediterranean dietary pattern in Scandinavian populations is low. In a cohort where few participants habitually consume olive oil, legumes, and fish in the combinations that define Mediterranean eating, the comparison group for "high meat consumers" is not people eating a Mediterranean diet — it is people eating less meat within a Northern European dietary context, which may look quite different nutritionally. This makes it difficult to use Mediterranean diet evidence as the interpretive frame for what the SNAC-K results mean in practice. It also underscores a broader point that our field has been slow to act on: dietary recommendations need to account for cultural and regional food environments. A precision nutrition approach for APOE4 carriers cannot simply export one population's dietary pattern to another; it must identify the underlying nutritional principles — adequate high-quality fat, omega-3 sufficiency, low processed food burden — and allow those to be achieved within the foods and traditions that people actually eat.
Take-Home Messages
The primary finding — a statistically significant APOE interaction for cognitive trajectories (P = .004) — is the paper’s substantive claim. The dementia interaction did not reach conventional significance (P = .10), which reflects limited power for that secondary endpoint rather than evidence against the cognitive finding.
The study is observational. Self-reported diet, a single Swedish cohort, and modest APOE34/44 subgroup sizes constrain interpretation. The triangulation design partially addresses reverse causation, but residual confounding cannot be excluded.
Processed and unprocessed red meat are not the same exposure. The cognitive benefit was most pronounced for unprocessed meat; processed meat increased dementia risk regardless of genotype. This distinction is often lost in coverage of this study.
There is biologically plausible support for APOE4-specific dietary fat responses. The APOE4 brain has impaired glucose metabolism and compensates by preferring fat as an energy substrate. Hanson and colleagues showed that an acute high-fat meal improved cognition and AD biomarkers in APOE4 carriers with MCI but worsened them in non-carriers — exactly the kind of genotype-specific response the SNAC-K data reflect at a population level.
This does not mean APOE4 carriers should eat more red meat or adopt a ketogenic diet. The fats that matter most for the APOE4 brain are heart-healthy good fats, obtained from fish and seafood, but also from unprocessed red meat or plant-based foods. Processed red meat increases dementia risk regardless of genotype and should be avoided.
Dietary patterns are more important than individual foods. No single food drives or prevents Alzheimer’s disease. One example of a healthy pattern is the Mediterranean-style pattern — rich in fish, vegetables, whole grains, and olive oil, with modest unprocessed animal protein — remains the best-supported framework for cognitive health, including in APOE4 carriers.
“Mediterranean pattern” evidence does not map cleanly onto a Swedish cohort. Few SNAC-K participants habitually follow a Mediterranean dietary pattern, so the high-meat comparison group is not eating Mediterranean — it is eating less meat within a Northern European food context. This limits cross-study interpretation and highlights the need for personalized, culturally adapted dietary guidance that identifies the underlying nutritional principles — adequate high-quality fat including red meat, omega-3 sufficiency, low processed food burden — and allows people to meet them through the foods and traditions they actually live with.
The right response to this paper is to design better trials, not to change dietary recommendations. Genotype-stratified randomized nutrition trials are urgently needed. This study makes a compelling case for them.
References
Norgren J, Carballo-Casla A, Grande G, et al. Meat Consumption and Cognitive Health by APOE Genotype. JAMA Network Open. 2026;9(3):e266489. doi:10.1001/jamanetworkopen.2026.6489
Yassine HN, Finch CE. APOE Alleles and Diet in Brain Aging and Alzheimer’s Disease. Front Aging Neurosci. 2020;12:150. doi:10.3389/fnagi.2020.00150
Yassine HN, et al. Nutrition State of Science and Dementia Prevention: Recommendations of the Nutrition for Dementia Prevention Working Group. Lancet Healthy Longevity. 2022;3(8):e501–e512.
Hanson AJ, Bayer JL, Baker LD, et al. Differential effects of meal challenges on cognition, metabolism and biomarkers for apolipoprotein E varepsilon4 carriers and adults with mild cognitive impairment. J Alzheimers Dis. 2015;48(1):205–218. doi:10.3233/jad-150218
Yassine HN, Braskie MN, Mack WJ, et al. Association of Docosahexaenoic Acid Supplementation With Alzheimer Disease Stage in Apolipoprotein E ε4 Carriers: A Review. JAMA Neurol. 2017;74(3):339–347. doi:10.1001/jamaneurol.2016.4899
Yassine HN, Croteau E, Rawat V, et al. DHA Brain Uptake and APOE4 Status: A PET Study With [1-11C]-DHA. Alzheimers Res Ther. 2017;9(1):23. doi:10.1186/s13195-017-0250-1



Observational studies are inherently limited — and that’s exactly why we should be cautious when different camps start declaring specific foods as “sacred” or universally beneficial.
The same type of data is often used to support completely opposing narratives. That alone should make us pause.
Maybe the more important question isn’t which food is “good” or “bad” — but under which physiological conditions a certain food becomes helpful, neutral, or even harmful.
Thanks for the very helpful analysis.
One thought about this study: We have fairly good evidence that homotaurine improves Alzheimer’s by several measures in APOE ε4 carriers (with an allele dose effect). Taurine also has neuroprotective effects, and a recent study (DOI:10.1016/j.biopha.2025.118527) showed that, in organoids derived from an APOE ε4/ε4 Alzheimer’s patient, taurine treatment attenuated Aβ accumulation and decreased tau phosphorylation.
What if taurine’s effects are, like those of homotaurine, also stronger in APOE ε4 carriers? Given that processed meat has much less taurine than non-processed meat, one might hypothesize that the presence of taurine in unprocessed meat is driving at least part of the effect seen in APOE ε4 carriers in the Karolinska study.