Hormone Replacement Therapy, Brain Health, and APOE4
Understanding the benefits, risks, and unanswered questions surrounding hormone therapy and cognitive aging.
The patient
A 63-year-old woman came to see me with questions about hormone replacement therapy (HRT) and brain health. She recently found out that she has two copies of APOE4.
She began using a transdermal estrogen patch with micronized progesterone at age 56, around the time she entered menopause. Like many women at that stage, she was experiencing hot flashes, night sweats, and disrupted sleep. Her gynecologist prescribed hormone therapy and her symptoms improved significantly.
She has now been on therapy for seven years without complications.
Recently, however, she began noticing changes in memory, occasionally forgetting names or pausing to remember why she walked into a room. Nothing dramatic, but enough to make her concerned.
She came to the clinic with three straightforward questions:
Is hormone therapy safe for the brain?
Can it prevent dementia?
How long should I stay on it?
These questions are becoming increasingly common as more women remain on hormone therapy longer and awareness of Alzheimer’s disease grows.
But answering them leads to a careful discussion on benefit versus risk.
A conversation with Dr. Howard Hodis
Shortly after seeing this patient, I discussed the case with Dr. Howard Hodis, Director of the Atherosclerosis Research Unit at the University of Southern California and one of the leading investigators studying the cardiovascular biology of menopausal hormone therapy.
Dr. Hodis has spent decades studying how estrogen interacts with vascular aging, particularly how the timing of hormone therapy relative to menopause may determine whether it helps or harms the cardiovascular system.
One of the key points from our conversation was that hormone therapy is often discussed as if it were a single treatment with a single risk profile.
Biologically, it is not.
The effects of estrogen depend on several factors:
when therapy begins
the health of the vascular system
the hormone formulation
the biology of the individual patient
And when the discussion shifts from the cardiovascular system to the brain, the uncertainty increases even further.
Why Hormone Therapy Exists
Hormone therapy was originally developed to treat symptoms of menopause.
As ovarian estrogen production declines, many women develop:
hot flashes
night sweats
sleep disruption
mood changes and depression
vaginal dryness
accelerated bone loss
Among available treatments, estrogen therapy remains the most effective therapy for vasomotor symptoms.
Hormone therapy also plays an important role in bone health. After menopause, bone loss accelerates rapidly. Roughly one in two women will experience an osteoporotic fracture during their lifetime. Hip fractures in particular can be devastating—about one-third of women die within a year, and many never fully recover functional independence.
Randomized trials show estrogen therapy reduces fractures by 30–50% in postmenopausal women.
For symptom relief and bone protection, the benefits of hormone therapy are well established.
The controversy arises when hormone therapy is discussed as a preventive therapy for chronic diseases, particularly cardiovascular disease and dementia.
Who Should Not Take Hormone Therapy
Although hormone therapy is highly effective for menopausal symptoms, it is not appropriate for everyone.
Most clinical guidelines recommend avoiding systemic estrogen therapy in women with certain conditions where risks may outweigh benefits.
These include:
Estrogen-dependent cancers, particularly breast cancer
A history of venous thromboembolism (blood clots) such as deep vein thrombosis or pulmonary embolism
Stroke or significant cardiovascular disease
Active liver disease
Unexplained vaginal bleeding
The concern in these situations is that estrogen can influence coagulation pathways, hormone-sensitive tissues, and vascular biology, potentially increasing the risk of recurrent disease.
For women with a history of breast cancer, systemic hormone therapy is generally avoided because of concerns that estrogen could stimulate hormone-sensitive tumor cells.
Similarly, women with a history of blood clots may have increased thrombotic risk with oral estrogen therapy, although some studies suggest that transdermal estrogen may carry lower clotting risk.
Importantly, these recommendations apply primarily to systemic hormone therapy. In some cases, local vaginal estrogen preparations may still be considered because systemic absorption is minimal.
As with many areas of menopause care, decisions often require individualized risk assessment and shared decision-making between patient and clinician.
The Cardiovascular Context
One key fact about women’s health is often overlooked:
Cardiovascular disease is the leading cause of death in women.
Before menopause, women experience significantly lower rates of coronary heart disease than men. After menopause, that protection declines.
This observation led researchers decades ago to propose the estrogen cardioprotective hypothesis.
Early observational studies suggested women using hormone therapy had 30–50% lower rates of coronary heart disease.
To test this hypothesis more rigorously, researchers launched one of the largest clinical trials ever conducted.
The Women’s Health Initiative
The Women’s Health Initiative (WHI) randomized more than 27,000 postmenopausal women to hormone therapy or placebo.
Initial results suggested increased risks of:
stroke
blood clots
breast cancer (with combined therapy)
These findings dramatically changed clinical practice.
However, an important detail emerged later: most participants were older, with an average age of 63, and had begun therapy more than a decade after menopause.
This raised a critical question:
Does the timing of hormone therapy matter?
The Timing Hypothesis
The timing hypothesis proposes that estrogen’s effects depend on the stage of vascular—and possibly other organ—health when therapy begins.
Early after menopause, when arteries are relatively healthy, estrogen appears to support vascular function through:
improved endothelial function
reduced LDL oxidation
improved vasodilation
reduced inflammation
But once atherosclerosis is established, estrogen may behave differently.
Experimental work suggests estrogen can increase matrix metalloproteinases such as MMP-9, enzymes involved in plaque instability.
In other words, estrogen may help prevent early vascular disease, but may not help—and could theoretically worsen—advanced disease.
Evidence for Timing
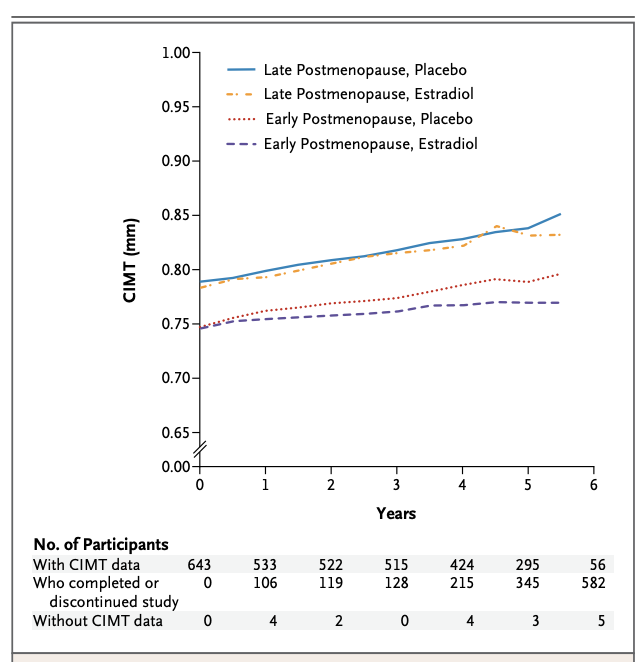
The ELITE trial (Early vs Late Intervention Trial with Estradiol) directly tested this hypothesis.
Women were randomized to estradiol or placebo and stratified according to time since menopause.
Women who began estradiol within six years of menopause showed slower progression of carotid atherosclerosis.
Women who began therapy more than ten years after menopause showed no benefit.
Figure. Effect of estradiol therapy on progression of carotid intima–media thickness (CIMT) according to time since menopause (ELITE Trial).
Mean carotid intima–media thickness (CIMT) as a measure of atherosclerosis measured over six years in women randomized to oral estradiol or placebo, stratified by time since menopause. In women in early postmenopause (<6 years since menopause), estradiol significantly slowed the progression of CIMT compared with placebo. In women in late postmenopause (≥10 years since menopause), estradiol did not reduce CIMT progression compared with placebo. These findings support the timing hypothesis, suggesting that the vascular effects of estrogen depend on when therapy is initiated relative to menopause. Data adapted from the Early vs Late Intervention Trial with Estradiol (ELITE)
Cardiovascular Outcomes
Meta-analyses of randomized trials have examined clinical outcomes.
Among women who started hormone therapy:
before age 60, or
within 10 years of menopause
studies show approximately:
30–40% reduction in coronary heart disease events
~30% reduction in all-cause mortality
A Danish randomized trial similarly found reduced cardiovascular events in recently postmenopausal women receiving hormone therapy for up to 16 years.
Hormone Therapy and the Brain
While much research has focused on cardiovascular disease, patients are often most concerned about brain health.
Estrogen influences several biological processes relevant to cognition:
synaptic plasticity
mitochondrial function
cerebral blood flow
inflammation
neuronal metabolism
Laboratory studies suggest estrogen may support neuronal resilience and synaptic function.
This biological rationale led many researchers to believe hormone therapy might help prevent Alzheimer’s disease.
However, clinical trials have not confirmed that expectation.
Hormone Therapy and Dementia Prevention
The most important trial examining this question was the Women’s Health Initiative Memory Study (WHIMS).
WHIMS enrolled more than 7,000 women aged 65 and older to test whether hormone therapy could prevent dementia.
The results were unexpected.
Women receiving combined estrogen-progestin therapy had approximately twice the risk of dementia compared with placebo when therapy was started after age 65.
Importantly, this finding does not take into account the role of timing.
Later studies suggest that hormone therapy begun earlier in life does not increase dementia risk, but it also has not convincingly reduced it.
At present, the conclusion is straightforward:
There is no strong randomized evidence that hormone therapy prevents dementia.
APOE4 and Brain Vulnerability
The picture becomes more complex when genetics are considered.
The APOE4 allele is the strongest common genetic risk factor for Alzheimer’s disease.
APOE4 carriers often show:
earlier amyloid deposition
greater vascular dysfunction
increased blood–brain barrier fragility
Importantly, not all APOE4 carriers are the same.
A cognitively normal APOE4 carrier may still have significant neural reserve.
But once someone develops mild cognitive impairment or dementia, the brain becomes far less resilient.
Estrogen, MMP-9, and Neurovascular Biology
One pathway linking estrogen to both cardiovascular and neurological disease involves matrix metalloproteinase-9 (MMP-9).
MMP-9 regulates:
extracellular matrix remodeling
vascular stability
blood–brain barrier permeability
Elevated MMP-9 activity has been associated with:
plaque rupture in cardiovascular disease
blood–brain barrier breakdown
neuroinflammation
Estrogen signaling can influence MMP-9 activity, suggesting that estrogen’s effects may differ depending on the stage of vascular or neurodegenerative disease.
The Problem of Uncertainty
Despite decades of research, hormone therapy remains one of the most debated topics in medicine.
Several factors contribute to this uncertainty.
Different studies examine different populations. Many randomized trials studied women who began therapy long after menopause, while clinical practice typically begins therapy earlier.
Hormone formulations also differ. Earlier trials studied oral conjugated estrogens and synthetic progestins, whereas modern regimens often use transdermal estradiol and micronized progesterone.
Most hormone trials were designed to study cardiovascular disease and fractures, not dementia.
And despite the major role of APOE genetics in Alzheimer’s disease, very few studies have stratified hormone therapy outcomes by genotype.
Current evidence on hormone therapy and dementia has important gaps, particularly for APOE4 carriers. Most randomized trials were not designed to study genetic subgroups and rarely stratified participants by APOE genotype. In addition, many trials initiated hormone therapy years after menopause, limiting their ability to test the timing hypothesis in the brain.
As a result, we still lack trials that start hormone therapy near menopause, follow women long-term, and specifically evaluate cognitive outcomes in APOE4 carriers. Studies using modern hormone formulations and neuroimaging or biomarker endpoints are also limited. These gaps mean that the effects of hormone therapy on dementia risk, especially in genetically vulnerable populations, remain uncertain.
Taken together, these limitations mean that hormone therapy decisions still require clinical judgment rather than simple rules.
Returning to the Patient
After discussing the science, the question comes back to the woman who started this conversation.
Like many medical interventions, the value of hormone therapy depends on several factors:
timing
biology
individual risk factors
patient goals
For my patient, the answer is not straightforward.
Hormone therapy likely helped her menopausal symptoms, and, given that she started treatment near menopause and has tolerated it well, it may still be reasonable to continue.
But when it comes to protecting the brain from dementia, science has not yet provided a definitive answer.
New memory concerns should prompt a formal cognitive assessment and a thoughtful discussion about whether continuing hormone therapy remains appropriate.
Take-Home Messages
Hormone therapy remains the most effective treatment for menopausal symptoms.
Timing matters. Starting hormone therapy near the menopausal transition appears biologically different from starting it decades later. Much of the risk seen in earlier trials occurred in women who initiated therapy long after menopause, highlighting the importance of timing.
Cardiovascular effects depend on vascular health. Estrogen may support vascular function in relatively healthy arteries, but its effects may differ once atherosclerosis is established, potentially altering plaque biology.
Hormone therapy is primarily a treatment for menopausal symptoms. Its strongest evidence supports relief of vasomotor symptoms and prevention of bone loss. Current evidence does not support using HRT as a general longevity or dementia-prevention therapy.
Cognitive symptoms warrant reassessment. When mild cognitive impairment or dementia emerges, there is less brain resilience, which may alter the risk–benefit balance, and continued therapy should be reassessed with a clinician.
Genetics may modify risk. Factors such as the APOE4 genotype, along with vascular health and baseline cognitive status, may influence how hormone therapy affects the brain, though this area remains incompletely studied.
Ultimately, hormone therapy should not be viewed as either a miracle therapy or a dangerous drug.
Until the science becomes clearer, the most important step is not a universal rule, but careful conversations between patients and their physicians, weighing symptoms, risks, and individual goals.



What I appreciated about this piece is that it takes a highly polarized topic and approaches it through the more nuanced lens of brain health, timing, risk, and individual variation. Framing hormone replacement therapy in relation to cognition and APOE4 is especially valuable, because it moves the discussion beyond simplistic “for or against” narratives and into the territory where clinical decision-making actually lives. That alone makes the post feel timely, thoughtful, and medically important. 
One nuance that could make the piece even stronger is to keep drawing a very explicit distinction between what is biologically plausible, what is supported by observational or subgroup data, and what is firmly established for routine practice. HRT, brain aging, and APOE4 sit at the intersection of real promise and real uncertainty, so readers benefit when the hierarchy of evidence is especially clear. That kind of calibration is not a weakness; it is exactly what makes complex prevention medicine more credible and more useful. 
Overall, I think this is a valuable and intellectually honest contribution to an area where many women are searching for more precise guidance. Thoughtful and important work!
Thank you for your fantastic work. Thorough yet concise.